|
As the table shows, the presence of the other oxidation states varies, but follows some patterns. 
Oxidation state 0 occurs for all elements – it is simply the element in its elemental form. With a chlorine ion (a chlorine atom that has gained one electron, Cl –), the oxidation number would be –1. These have oxidation numbers of +2 & +3 respectively. Similarly, iron (Fe) can lost two electrons to form the Fe 2+ ion, or lose three electrons to form the Fe 3+ ion. These have an oxidation state of +1, the same as the charge on the ion. For example, sodium (Na) can lose an electron to form sodium ions (Na +). If your only experience of chemistry is from back in school, you might remember the concept of ions – atoms that have gained or lost electrons to form positively or negatively charged ions. The sum of all of the oxidation states of the various elements in a compound should be zero. Similarly, adding electrons results in a negative oxidation state. Removing negative charges from an atom results, therefore, in a positive oxidation state. This might initially seem counter-intuitive, but remember that electrons are negatively charged. An element that is not combined with any other different elements has an oxidation state of 0, because it has had no electrons added or removed.Īn atom of an element in a compound will have a positive oxidation state if it has had electrons removed. It’s essentially a number assigned to an element in a compound, and (at a basic level) denotes the number of electrons that have been either removed from or added to that element. To try to solve that, I made the table you can see at the top of the post here.įor non-chemists, it’s probably worth clarifying exactly what we mean by ‘oxidation state’. This is actually quite an easy concept for chemists to use, but a tricky one to define. I was looking for one of these last week, out of interest, and couldn’t find one that presented the information in an easy-to-comprehend manner. Today’s post is another variation on the theme: a periodic table showing the possible and common oxidation states of every element. You can effortlessly find every single detail about the elements from this single Interactive Periodic table.There are a whole host of periodic tables out there, and I’ve already added to the pile with a periodic table of data previously. Let me tell you how this Interactive Periodic Table will help you in your studies.ġ). Number Elements Valence electrons 1 Hydrogen (H)Ģ 21 Scandium (Sc) 3 22 Titanium (Ti) 4 23 Vanadium (V) 5 24 Chromium (Cr) 6 25 Manganese (Mn) 7 26 Iron (Fe) 8 27 Cobalt (Co) 9 28 Nickel (Ni) 10 29 Copper (Cu) 11 30 Zinc (Zn) 12 31 Gallium (Ga) 3 32 Germanium (Ge) 4 33 Arsenic (As) 5 34 Selenium (Se) 6 35 Bromine (Br) 7 36 Krypton (Kr) 8 37 Rubidium (Rb) 1 38 Strontium (Sr) 2 39 Yttrium (Y) 3 40 Zirconium (Zr) 4 41 Niobium (Nb) 5 42 Molybdenum (Mo) 6 43 Technetium (Tc) 7 44 Ruthenium (Ru) 8 45 Rhodium (Rh) 9 46 Palladium (Pd) 10 47 Silver (Ag) 11 48 Cadmium (Cd) 12 49 Indium (In) 3 50 Tin (Sn) 4 51 Antimony (Sb) 5 52 Tellurium (Te) 6 53 Iodine (I) 7 54 Xenon (Xe) 8 55 Cesium (Cs) 1 56 Barium (Ba) 2 57 Lanthanum (La) 3 58 Cerium (Ce) 4 59 Praseodymium (Pr) 5 60 Neodymium (Nd) 6 61 Promethium (Pm) 7 62 Samarium (Sm) 8 63 Europium (Eu) 9 64 Gadolinium (Gd) 10 65 Terbium (Tb) 11 66 Dysprosium (Dy) 12 67 Holmium (Ho) 13 68 Erbium (Er) 14 69 Thulium (Tm) 15 70 Ytterbium (Yb) 16 71 Lutetium (Lu) 3 72 Hafnium (Hf) 4 73 Tantalum (Ta) 5 74 Tungsten (W) 6 75 Rhenium (Re) 7 76 Osmium (Os) 8 77 Iridium (Ir) 9 78 Platinum (Pt) 10 79 Gold (Au) 11 80 Mercury (Hg) 12 81 Thallium (Tl) 3 82 Lead (Pb) 4 83 Bismuth (Bi) 5 84 Polonium (Po) 6 85 Astatine (At) 7 86 Radon (Rn) 8 87 Francium (Fr) 1 88 Radium (Ra) 2 89 Actinium (Ac) 3 90 Thorium (Th) 4 91 Protactinium (Pa) 5 92 Uranium (U) 6 93 Neptunium (Np) 7 94 Plutonium (Pu) 8 95 Americium (Am) 9 96 Curium (Cm) 10 97 Berkelium (Bk) 11 98 Californium (Cf) 12 99 Einsteinium (Es) 13 100 Fermium (Fm) 14 101 Mendelevium (Md) 15 102 Nobelium (No) 16 103 Lawrencium (Lr) 3 104 Rutherfordium (Rf) 4 105 Dubnium (Db) 5 106 Seaborgium (Sg) 6 107 Bohrium (Bh) 7 108 Hassium (Hs) 8 109 Meitnerium (Mt) 9 110 Darmstadtium (Ds) 10 111 Roentgenium (Rg) 11 112 Copernicium (Cn) 12 113 Nihonium (Nh) 3 114 Flerovium (Fl) 4 115 Moscovium (Mc) 5 116 Livermorium (Lv) 6 117 Tennessine (Ts) 7 118 Oganesson (Og) 8 Free Gift for you: Interactive Periodic Table Valence Electrons Chart for All Elements Atomic 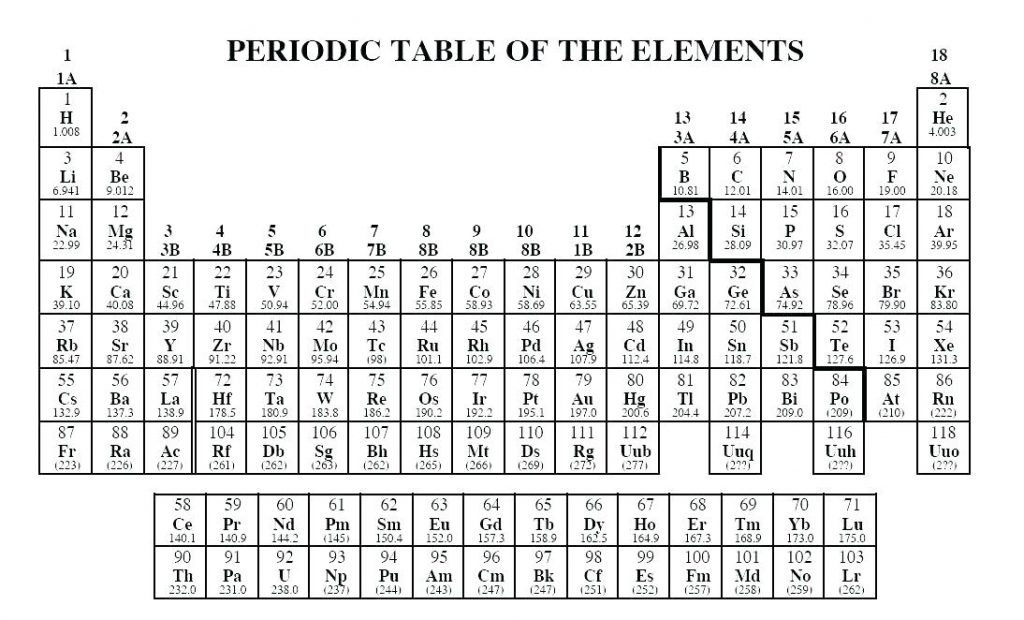
(Where you will get the HD images along with the explanation). If you want a Periodic table with Valence electrons, then visit Periodic table with Valence electrons labeled in it. 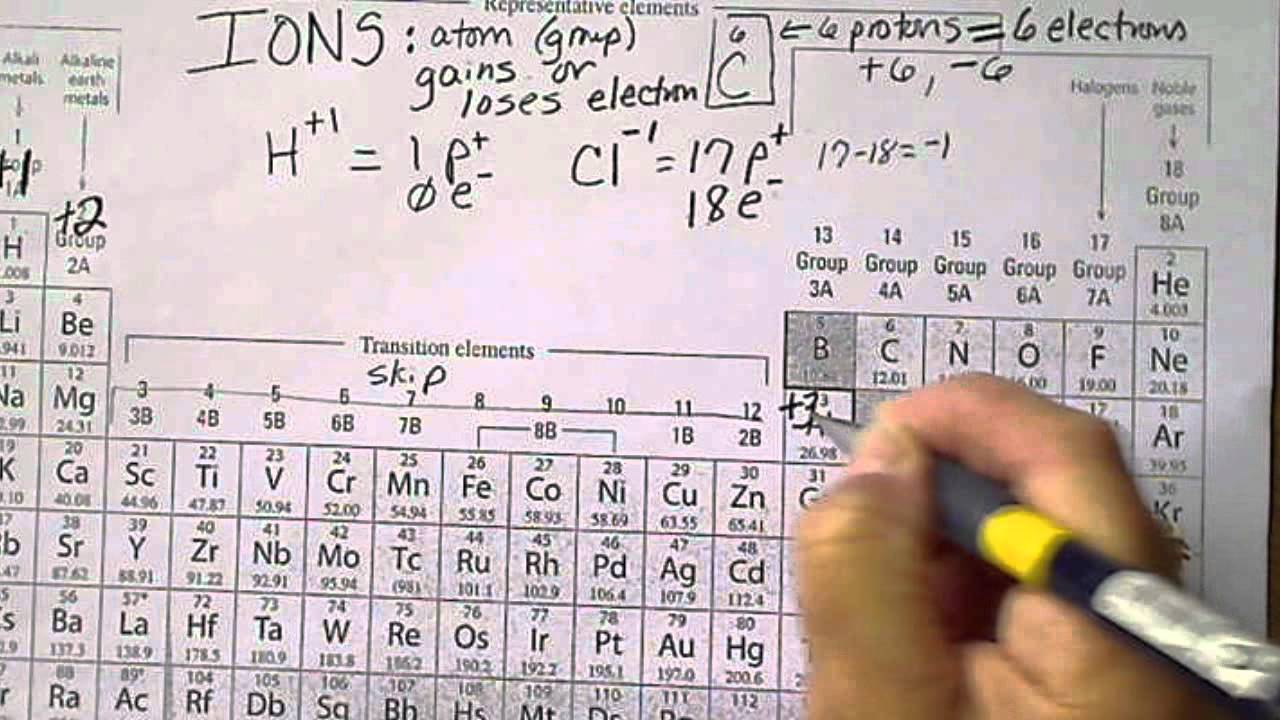
But for most of the transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core.For main group elements (i.e s-block and p-block elements), the valence electrons are the electrons present in the outermost orbit.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
